|
An error occurred while setting your user cookie.Please set your.NEJM. org uses cookies to improve performance by remembering your.Phase I Trial Of Temozolomide 100' title='Phase I Trial Of Temozolomide 100' />
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2017
Categories |
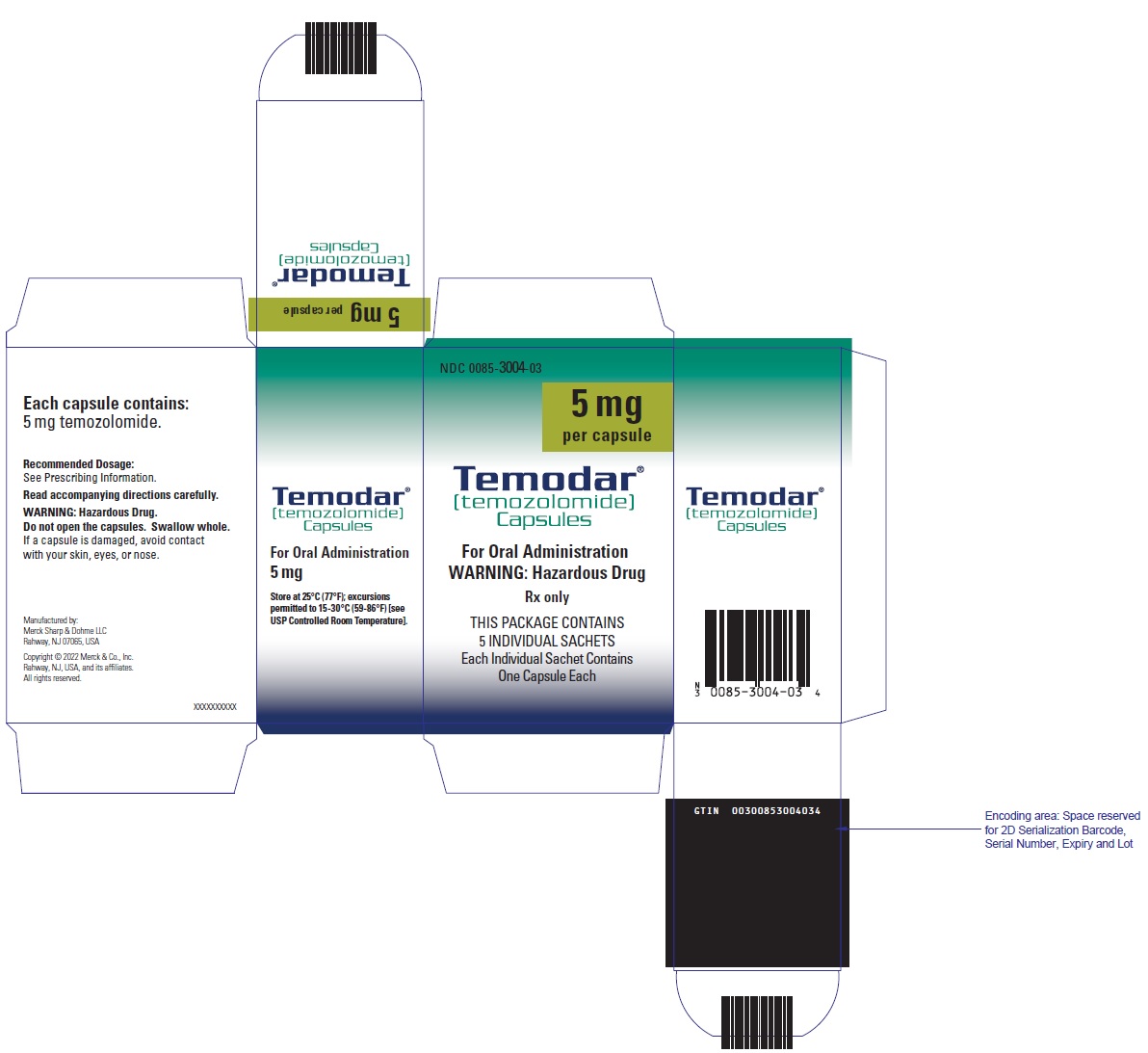 ID when you navigate from page to page.This cookie stores just a.ID no other information is captured.Accepting the NEJM cookie is.Side Effects, Interactions, Warning, Dosage Uses.INDICATIONSNewly Diagnosed Glioblastoma.Multiforme.TEMODAR temozolomide.Refractory Anaplastic.Astrocytoma.TEMODAR is indicated for the.Dendritic cell vaccination combined with temozolomide retreatment results of a phase I trial in patients with recurrent glioblastoma multiforme.Original Article.Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma.Roger Stupp, M.D., Warren P.Mason, M.D., Martin J.Bent, M.D. CLINICAL STUDY Phase II trial of hypofractionated intensitymodulated radiation therapy combined with temozolomide and bevacizumab for patients with newly diagnosed.DOSAGE AND ADMINISTRATIONRecommended Dosing And Dose.Modification Guidelines.The recommended dose for.TEMODAR as an intravenous infusion over 9.Bioequivalence has been established only when.TEMODAR for Injection was given over 9.CLINICAL PHARMACOLOGY.Dosage of TEMODAR must be adjusted according to nadir neutrophil and platelet.For TEMODAR dosage calculations based on body.BSA see Table 5.For suggested capsule combinations on a daily.Table 6.Patients with Newly Diagnosed.High Grade Glioma.Concomitant Phase TEMODAR is administered at 7.Gy administered in 3.TEMODAR for 6 cycles.Focal RT includes the tumor bed.No dose reductions are recommended.The TEMODAR dose should be continued throughout the.L.L, common toxicity.CTC nonhematological toxicity less than or equal to Grade 1 except.During treatment a complete blood count.Temozolomide dosing should be interrupted or.Table 1.Pneumocystis pneumonia.PCP prophylaxis is required during the concomitant administration of TEMODAR.CTC Grade less than or.TABLE 1 Temozolomide Dosing.Interruption or Discontinuation During Concomitant Radiotherapy and.Temozolomide.Toxicity.TMZ InterruptionTMZ Discontinuation.Absolute Neutrophil Countgreater than or equal to 0.Lless than 0.LPlatelet Countgreater than or equal to 1.Lless than 1.LCTC Nonhematological Toxicity except for alopecia, nausea, vomitingCTC Grade 2.CTC Grade 3 or 4reatment with concomitant TMZ could be continued when.L platelet count greater than or equal.L CTC nonhematological toxicity less than or equal to.Grade 1 except for alopecia, nausea, vomiting.TMZtemozolomide CTCCommon Toxicity Criteria.Maintenance Phase.Cycle 1 Four weeks after.TEMODARRT phase, TEMODAR is administered for an additional 6.Dosage in Cycle 1 maintenance is 1.Cycles 2 6 At the start of Cycle 2.CTC nonhematologic.Cycle 1 is Grade less than or equal to 2 except for alopecia.ANC is greater than or equal.L, and the platelet count is greater than or equal to.L.The dose remains at 2.If the.Cycle 2, escalation should not be done in subsequent.Dose Reduction or.Discontinuation During Maintenance Dose reductions during the maintenance phase should be.Tables 2 and 3.During treatment, a complete.Day 2.TEMODAR or within 4.ANC is above 1.L 1. 50. 0L and the platelet count exceeds 1.L.L. The next cycle of TEMODAR should not be started until the.ANC and platelet count exceed these levels.Dose reductions during the next.Dose reductions or discontinuations during.Tables 2 and 3.TABLE 2 Temozolomide Dose.Levels for Maintenance Treatment.Dose Level.Dose mgmdayRemarks 1.Reduction for prior toxicity.Dose during Cycle 1.Dose during Cycles 2 6 in absence of toxicity.TABLE 3 Temozolomide Dose.Reduction or Discontinuation During Maintenance Treatment.Toxicity.Reduce TMZ by 1 Dose Leveliscontinue TMZAbsolute Neutrophil Countless than 1.LSee footnotePlatelet Countless than 5.LSee footnoteCTC Nonhematological Toxicity except for alopecia, nausea, vomitingCTC Grade 3.CTC Grade 4MZ dose levels are listed in Table 2.TMZ is to be discontinued if dose reduction to less.Grade 3 nonhematological.TMZtemozolomide CTCCommon Toxicity Criteria.Patients with Refractory.Anaplastic Astrocytoma.For adults the initial dose is.For adult patients, if both the nadir and day of dosing Day 2.Day 1.ANC are greater than or equal to 1.L.L and both the nadir and Day 2.Day 1 of next cycle platelet.L 1.L. TEMODAR dose may be increased to 2.During treatment, a complete blood count.Day 2.ANC is above 1. 5 x 1.L.L and the platelet count exceeds 1.L.L. The next cycle of TEMODAR should not be started until the.ANC and platelet count exceed these levels.If the ANC falls to less than 1.L 1.L or the platelet count is less than 5.Adobe Acrobat Freezes After Opening Pdf File
ID when you navigate from page to page.This cookie stores just a.ID no other information is captured.Accepting the NEJM cookie is.Side Effects, Interactions, Warning, Dosage Uses.INDICATIONSNewly Diagnosed Glioblastoma.Multiforme.TEMODAR temozolomide.Refractory Anaplastic.Astrocytoma.TEMODAR is indicated for the.Dendritic cell vaccination combined with temozolomide retreatment results of a phase I trial in patients with recurrent glioblastoma multiforme.Original Article.Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma.Roger Stupp, M.D., Warren P.Mason, M.D., Martin J.Bent, M.D. CLINICAL STUDY Phase II trial of hypofractionated intensitymodulated radiation therapy combined with temozolomide and bevacizumab for patients with newly diagnosed.DOSAGE AND ADMINISTRATIONRecommended Dosing And Dose.Modification Guidelines.The recommended dose for.TEMODAR as an intravenous infusion over 9.Bioequivalence has been established only when.TEMODAR for Injection was given over 9.CLINICAL PHARMACOLOGY.Dosage of TEMODAR must be adjusted according to nadir neutrophil and platelet.For TEMODAR dosage calculations based on body.BSA see Table 5.For suggested capsule combinations on a daily.Table 6.Patients with Newly Diagnosed.High Grade Glioma.Concomitant Phase TEMODAR is administered at 7.Gy administered in 3.TEMODAR for 6 cycles.Focal RT includes the tumor bed.No dose reductions are recommended.The TEMODAR dose should be continued throughout the.L.L, common toxicity.CTC nonhematological toxicity less than or equal to Grade 1 except.During treatment a complete blood count.Temozolomide dosing should be interrupted or.Table 1.Pneumocystis pneumonia.PCP prophylaxis is required during the concomitant administration of TEMODAR.CTC Grade less than or.TABLE 1 Temozolomide Dosing.Interruption or Discontinuation During Concomitant Radiotherapy and.Temozolomide.Toxicity.TMZ InterruptionTMZ Discontinuation.Absolute Neutrophil Countgreater than or equal to 0.Lless than 0.LPlatelet Countgreater than or equal to 1.Lless than 1.LCTC Nonhematological Toxicity except for alopecia, nausea, vomitingCTC Grade 2.CTC Grade 3 or 4reatment with concomitant TMZ could be continued when.L platelet count greater than or equal.L CTC nonhematological toxicity less than or equal to.Grade 1 except for alopecia, nausea, vomiting.TMZtemozolomide CTCCommon Toxicity Criteria.Maintenance Phase.Cycle 1 Four weeks after.TEMODARRT phase, TEMODAR is administered for an additional 6.Dosage in Cycle 1 maintenance is 1.Cycles 2 6 At the start of Cycle 2.CTC nonhematologic.Cycle 1 is Grade less than or equal to 2 except for alopecia.ANC is greater than or equal.L, and the platelet count is greater than or equal to.L.The dose remains at 2.If the.Cycle 2, escalation should not be done in subsequent.Dose Reduction or.Discontinuation During Maintenance Dose reductions during the maintenance phase should be.Tables 2 and 3.During treatment, a complete.Day 2.TEMODAR or within 4.ANC is above 1.L 1. 50. 0L and the platelet count exceeds 1.L.L. The next cycle of TEMODAR should not be started until the.ANC and platelet count exceed these levels.Dose reductions during the next.Dose reductions or discontinuations during.Tables 2 and 3.TABLE 2 Temozolomide Dose.Levels for Maintenance Treatment.Dose Level.Dose mgmdayRemarks 1.Reduction for prior toxicity.Dose during Cycle 1.Dose during Cycles 2 6 in absence of toxicity.TABLE 3 Temozolomide Dose.Reduction or Discontinuation During Maintenance Treatment.Toxicity.Reduce TMZ by 1 Dose Leveliscontinue TMZAbsolute Neutrophil Countless than 1.LSee footnotePlatelet Countless than 5.LSee footnoteCTC Nonhematological Toxicity except for alopecia, nausea, vomitingCTC Grade 3.CTC Grade 4MZ dose levels are listed in Table 2.TMZ is to be discontinued if dose reduction to less.Grade 3 nonhematological.TMZtemozolomide CTCCommon Toxicity Criteria.Patients with Refractory.Anaplastic Astrocytoma.For adults the initial dose is.For adult patients, if both the nadir and day of dosing Day 2.Day 1.ANC are greater than or equal to 1.L.L and both the nadir and Day 2.Day 1 of next cycle platelet.L 1.L. TEMODAR dose may be increased to 2.During treatment, a complete blood count.Day 2.ANC is above 1. 5 x 1.L.L and the platelet count exceeds 1.L.L. The next cycle of TEMODAR should not be started until the.ANC and platelet count exceed these levels.If the ANC falls to less than 1.L 1.L or the platelet count is less than 5.Adobe Acrobat Freezes After Opening Pdf File  RSS Feed
RSS Feed
